Blog
May 22, 2024
Comparing Endoscope Testing Solutions: How Dovideq’s LightControl Stands Out
May 10, 2023
In the realm of healthcare, the safe and effective reuse of medical devices, particularly endoscopes, is critical. ISO...
April 24, 2023
Endoscopes are crucial tools in modern medicine, allowing doctors to perform a wide range of procedures inside the body...
March 14, 2023
Endoscopes are vital medical devices that enable doctors to diagnose and treat a variety of conditions by providing a...
November 11, 2022
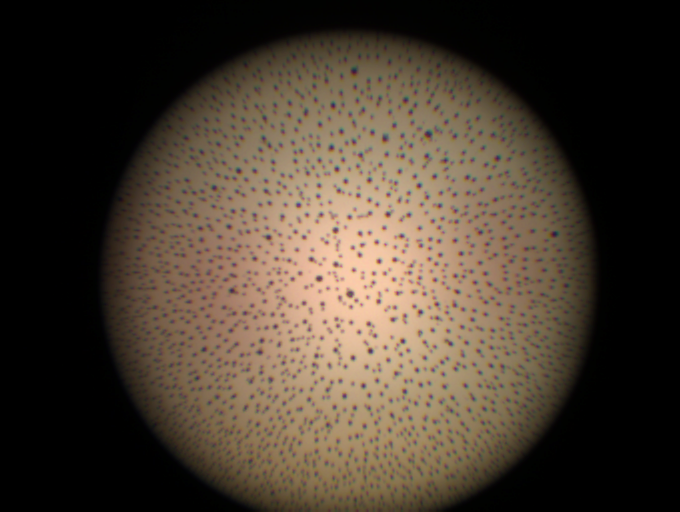
"Have you ever wondered what the best way is to inspect a fiber optic cable?" Everyone knows that when it comes to...
dovideqmedical,
Dovideq,
medical,
cost saving,
medical cost saving,
medical cost reduction,
endoscope,
cost,
cost reduction,
endoscope testing,
test tag,
MDR,
sterilization,
Department,
hospital,
CSSD,
sterilization department,
sterilisation,
about us,
MDR2017/745,
compliancy,
audit,
repair
October 25, 2022
January 31, 2022
Regulatory framework With the introduction of Regulation EU MDR 2017/745, reusable rigid endoscopes are now class I...
January 26, 2022
It may not sound like much fun to keep track of your endoscope repair company, but since you're handing over your...
December 20, 2021
All (supra)national regulatory authorities require quality control of endoscopes before each reuse. In addition, a...